Accelerating interest in silicon nitride thin film material system continues in both academic and industrial communities due to its highly desirable physical, chemical, and electrical properties and the potential to enable new device technologies. As considered here, the silicon nitride material system encompasses both non-hydrogenated (SiNx) and hydrogenated (SiNx:H) silicon nitride, as well as silicon nitride-rich films, defined as SiNx with C inclusion, in both non-hydrogenated (SiNx(C)) and hydrogenated (SiNx:H(C)) forms. Due to the extremely high level of interest in these materials, this article is intended as a follow-up to the authors' earlier publication [A. E. Kaloyeros, F. A. Jové, J. Goff, B. Arkles, Silicon nitride and silicon nitride-rich thin film technologies: trends in deposition techniques and related applications, ECS J. Solid State Sci. Technol., 6, 691 (2017)] that summarized silicon nitride research and development (R&D) trends through the end of 2016. In this survey, emphasis is placed on cutting-edge achievements and innovations from 2017 through 2019 in Si and N source chemistries, vapor phase growth processes, film properties, and emerging applications, particularly in heterodevice areas including sensors, biointerfaces and photonics.

The Electrochemical Society (ECS) was founded in 1902 to advance the theory and practice at the forefront of electrochemical and solid state science and technology, and allied subjects.
ISSN: 2162-8777
JSS is a peer-reviewed journal covering fundamental and applied areas of solid-state science and technology, including experimental and theoretical aspects of the chemistry, and physics of materials and devices.
Open all abstracts, in this tab
Alain E. Kaloyeros et al 2020 ECS J. Solid State Sci. Technol. 9 063006
Roy Knechtel et al 2021 ECS J. Solid State Sci. Technol. 10 074008
Wafer bonding is an important process step in microsystem technologies for processing engineered substrates and for capping. Usually, the work and literature are focused on the bonding of the main wafer area. However, in recent years MEMS technologies have become more complex, with more process steps after wafer bonding. Accordingly, the wafer edge is becoming more and more important, and must be engineered. Methods for realizing this are discussed in this paper.
Sandeep Arya et al 2021 ECS J. Solid State Sci. Technol. 10 023002
ZnO has several potential applications into its credit. This review article focuses on the influence of processing parameters involved during the synthesis of ZnO nanoparticles by sol-gel method. During the sol-gel synthesis technique, the processing parameters/experimental conditions can affect the properties of the synthesized material. Processing parameters are the operating conditions that are to be kept under consideration during the synthesis process of nanoparticles so that various properties exhibited by the resulting nanoparticles can be tailored according to the desired applications. Effect of parameters like pH of the sol, additives used (like capping agent, surfactant), the effect of annealing temperature and calcination on the morphology and the optical properties of ZnO nanoparticles prepared via sol-gel technique is analyzed in this study. In this study, we tried to brief the experimental investigations done by various researchers to analyze the influence of processing parameters on ZnO nanoparticles. This study will provide a platform to understand and establish a correlation between the experimental conditions and properties of ZnO nanoparticles prepared through sol-gel route which will be helpful in meeting the desired needs in various application areas.
Kartika A. Madurani et al 2020 ECS J. Solid State Sci. Technol. 9 093013
Graphene is a thin layer carbon material that has become a hot topic of research during this decade due to its excellent thermal conductivity, mechanical strength, current density, electron mobility and surface area. These extraordinary properties make graphene to be developed and applied in various fields. On this basis, researchers are interested to find out the methods to produce high quality graphene for industrial use. Various methods have been developed and reported to produce graphene. This paper was designed to summarize the development of graphene synthesis methods and the properties of graphene products that were obtained. The application of graphene in the various fields of environment, energy, biomedical, sensors, bio-sensors, and heat-sink was also summarized in this paper. In addition, the history, challenges, and prospects of graphene production for research and industrial purposes were also discussed.
Yu-Cheng Syu et al 2018 ECS J. Solid State Sci. Technol. 7 Q3196
Biosensor research has been addressed as an interested field recently. Within different kinds of developed biosensing technologies, field-effect transistor (FET) based biosensors stand out due to their attractive features, such as ultra-sensitivity detection, mass-production capability, and low-cost manufacturing. To promote understandings of the FET based biosensing technology, in this review, its sensing mechanism is introduced, as well as major FET-based biosensing devices: ion sensitive field-effect transistor (ISFET), silicon nanowire, organic FET, graphene FET, and compound-semiconductor FET. In addition to FET-based biosensing devices, clinical applications, such as cardiovascular diseases (CVDs), cancers, diabetes, HIV, and DNA sequence, are also reviewed. In the end, several critical challenges of FET-based biosensing technology are discussed to envision next steps in healthcare technologies.
Chunlin Zhou et al 2021 ECS J. Solid State Sci. Technol. 10 027005
In recent years, betavoltaic batteries have become an ideal power source for micro electromechanical systems. Betavoltaic battery is a device that converts the decay energy of beta emitting radioisotope sources into electrical energy using transducers. They have the advantages of high energy density, long service life, strong anti-interference ability, small size, light weight, easy miniaturization and integration, thus it has become a research hotspot in the field of micro energy. However, to date, the low energy conversion efficiencies as well as technological limitations of betavoltaic batteries impede their further application. In this review, the theory of betavoltaic energy conversion and recent understanding of the ideal material and structure design of the betavoltaic batteries for efficient exciton production, dissociation and charge transport is described, as well as recent attempts to realize optimum results. This review article concludes by identifying the remaining challenges for the improvement of battery performance and by providing perspectives toward real application of betavoltaic batteries.
Alain E. Kaloyeros and Barry Arkles 2024 ECS J. Solid State Sci. Technol. 13 043001
Silicon carbide (SiCx) thin films deposition processes fall primarily into three main categories: (1) chemical vapor deposition (CVD) and its variants, including plasma enhanced CVD (PE-CVD); (2) physical vapor deposition (PVD), including various forms of sputtering; (3) alternative (non-CVD and non-PVD) methodologies. Part I of this two-part report ECS J. Solid State Sci. Technol., 12, 103001 (2023) examined recent peer-reviewed publications available in the public domain pertaining to the various CVD processes for SiCx thin films and nanostructures, as well as CVD modeling and mechanistic studies. In Part II, we continue our detailed, systematic review of the latest progress in cutting-edge SiCx thin film innovations, focusing on PVD and other non-PVD and non-CVD SiCx coating technologies. Particular attention is given to pertinent experimental details from PVD and alternative (non-CVD and non-PVD) processing methodologies as well as their influence on resulting film properties and performance.
Jose Lopez Ninantay et al 2024 ECS J. Solid State Sci. Technol. 13 054004
The catalytic decomposition of poly(phthalaldehyde) with a photoacid generator can be used as dry-develop photoresist, where the exposed film depolymerizes into small molecules to allow the development of features via controlled vaporization. Higher temperatures enabled shorter dry-development times, but also promoted faster photoacid diffusion that compromised pattern fidelity. Trihexylamine was used as a base quencher to counteract acid diffusion in a phthalaldehyde-propanal co-polymer photoresist. The propanal co-monomer in the polymer improves the vaporization rate because it has a higher vapor pressure than phthalaldehyde. Addition of the base quencher was found to improve the contrast, pattern fidelity, and ease-of-handling of the dry-develop resist in a direct-write UV lithography tool. The dry-development of 4 μm features was achieved with no appreciable residue. For large area features, a spatially variable exposure method was used to direct the residue away from the exposed area. The gradient exposure method was used to produce 100 μm features. Plasma etching after dry-development was also used to achieve residue-free dry-developed patterns. These results show the benefits of incorporating base additives into a dry-develop depolymerizable resist system and highlight the need for addressing residue formation.
Marko J. Tadjer et al 2019 ECS J. Solid State Sci. Technol. 8 Q3187
Gallium oxide (β-Ga2O3) is an emerging semiconductor with relevant properties for power electronics, solar-blind photodetectors, and some sensor applications due to its ultra-wide bandgap and developing technology base for high quality, melt-based substrate growth and thick, low-doped homoepitaxial layers. Of critical importance for the commercialization of this potentially important material is understanding of doping mechanisms in the monoclinic lattice, where two types of Ga sites and three types of O sites have been identified. A critical literature review of doping and defects of the monoclinic β-phase of gallium oxide is provided in this work. Theoretical fundamentals of both donor and acceptor doping in Ga2O3 are reviewed. Advances in doping of epitaxial Ga2O3 with a focus on molecular beam epitaxy and ion implantation are critically examined. As doping is fundamentally related to defects, particularly in this material, a review of defect characterization by optical and electrical spectroscopic methods is provided as well. P-type doping, one of the fundamental challenges for Ga2O3, is discussed in terms of first-principles calculations and ion implantation of known acceptors such as Mg and N.
Sean W. King 2015 ECS J. Solid State Sci. Technol. 4 N3029
Over the past decade, the primary focus for improving the performance of nano-electronic metal interconnect structures has been to reduce the impact of resistance-capacitance (RC) delays via utilizing insulating dielectrics with ever lower values of dielectric permittivity. The integration and implementation of such low dielectric constant (i.e. low-k) materials has been fraught with numerous challenges. For intermetal and interlayer (ILD) low-k dielectrics, these challenges have been largely associated to integration with metal interconnect fabrication processes and well documented and reviewed in the literature. Although equally important, less attention has been given to other low-k dielectrics utilized in metal interconnect structures that are commonly referred to as low-k dielectric barriers (DB), etch stops (ES), and/or Cu capping layers (CCL). These materials present numerous challenges as well for integration into metal interconnect fabrication processes. However, they also have more stringent integrated functionality requirements relative to low-k ILD materials that serve only a basic purpose of electrically isolating adjacent metal lines. In this article, we review the integration challenges and associated integrated functionality requirements for low-k DB/ES/CCL materials with a focus on the current status and future direction needed for these materials to facilitate both Moore's law (i.e. More Moore) and More than Moore scaling.
Open all abstracts, in this tab
M. M. Abdelhamied et al 2024 ECS J. Solid State Sci. Technol. 13 063003
In this research, the casting solution manufacturing approach was used to mix polyvinyl alcohol (PVA) and copper oxide (CuO) to create the composite (PVA/CuO). X-ray diffraction analysis and Fourier transform infrared spectroscopy were applied to record the successful fabrications of the composites. Next, argon ion beams at fluencies of 2.5 × 1017, 5 × 1017, and 7.5 × 1017 ions.cm−2 were used to irradiate the composites. In frequencies of 50 Hz to 6 MHz, the dielectric characteristics of PVA/CuO were modified by the ion irradiation. The dielectric constant was enhanced from 39 for unmodified PVA/CuO to 356 for the irradiated composite by 7.5 × 1017 ions.cm−2, and the conductivity changed from 0.05 × 10−6 S cm−1 to 2.9 × 10−6 S cm−1. However, the potential barrier decreased from 0.24 eV for PVA/CuO to 0.21, 0.16, and 0.15 eV, respectively, for 2.5 × 1017, 5 × 1017, and 7.5 × 1017 ions.cm−2, and the relaxation time decreased from 9.36 × 10−8 sec for PVA/CuO, to 6.58 × 10−8 sec for 7.5 × 1017 ions.cm−2. The results indicate that the irradiated PVA/CuO nanocomposite can be used in a number of devices such as capacitors and batteries.
C. Jenkins et al 2024 ECS J. Solid State Sci. Technol. 13 065003
In this study, pillar shaped yttria-stabilized zirconia (YSZ) 3D microstructures with ∼60 to 90 μm diameter and 12 to 20 μm height are fabricated by 3D inkjet printing to improve the topology of the electrolyte/cathode interface. The microstructures increase the surface area of the cell by ∼2.4% to 4.0% and enhance the connection between the dense YSZ electrolyte and mixed YSZ-lanthanum strontium manganite (LSM) cathode. The morphology and microstructure of the YSZ interface are characterized with scanning electron microscopy. Polarization curves confirm that the power density improves by 47% to 107% at 0.55 V, depending on the dimensions of the microstructures, in comparison to a flat interface. The non-linear improvement in power density with the size of microstructures is confirmed by calculating the uncertainty with repeated tests. Based on electrochemical impedance spectroscopy and distribution of relaxation times analysis, the performance improvement is attributed to changes in the oxygen surface exchange kinetics and O2− diffusivity in the cathode.
Naseem Akhtar et al 2024 ECS J. Solid State Sci. Technol. 13 064001
Here we present the fabrication of a multilayer resistive memory device (ReRAM) utilizing AlCr2O4/MXene nanocomposite. Comprehensive investigations into the structural and morphological properties of the nanostructures were conducted using various characterization techniques. The fabricated device was tested by measuring I-V characteristics at different current applications which encompasses all previous results. The band gap value for the nanocomposite was reduced to 2.42 eV while that for AlCr2O4 was measured at 3.25 eV via photoluminescence spectrum. Average particle size of the AlCr2O4/MXene nanocomposite was determined to be 25 nm through powder X-ray diffraction analysis. Crystallographic analysis revealed that all crystal peaks conform to the R-3c (167) space group, indicative of a standard hexagonal crystal structure. Energy-dispersive X-ray readings provided further confirmation that all required elements are present in the sample, affirming successful synthesis of the nanocomposite. Notably, the nanocomposite demonstrated exceptional performance as an electrode material in ReRAM, as evidenced by its current-voltage characteristics, making the AlCr2O4/MXene nanocomposite suitable for a wide range of next-generation device applications.
Chenliang Huo 2024 ECS J. Solid State Sci. Technol. 13 065002
Silicon has garnered significant attention as the primary material for solar cell preparation. Traditional alkaline etching solutions are limited to creating an upright pyramid structure on monocrystalline silicon surfaces. However, research indicates that an inverted pyramid structure exhibits superior light-trapping properties compared to the upright pyramid structure. In this study, we employed a one-step copper ion metal-assisted chemical etching process to fabricate an inverted pyramid structure on monocrystalline silicon wafers. This method allows for the customization of either inverted or upright pyramid structures by adjusting the concentration of specific solution components. Characterization of the textured silicon wafers reveals that the inverted pyramid structure exhibits lower reflectivity than both the upright pyramid structure and polished silicon. By integrating this texturing technique into the solar cell production line, we successfully produced solar cells with both inverted and upright pyramid structures. Evaluation of various solar cell parameters demonstrates that the inverted pyramid structure outperforms the upright pyramid structure, showcasing lower reflectivity and higher photoelectric conversion efficiency.
Xueyin Su et al 2024 ECS J. Solid State Sci. Technol. 13 065001
Defects induced by the source/drain process have a significant impact on the scattering mechanism of PMOS at cryogenic temperatures. Here, the cryogenic characteristics of FD-SOI devices with heavily doped epitaxial source/drain (Epi FD-SOI devices) and metallic Schottky barrier source/drain (SB FD-SOI devices) were investigated from 300 K down to 6 K. The doping profile along the channel was analyzed by TCAD simulation analysis. Experimental comparison of transistor performance at cryogenic temperatures was carried out for these devices with gate lengths (LG) of 100 nm and 40 nm. The I-V characteristics of the FD-SOI devices were measured with a liquid helium cooling environment. The cryogenic effect of the two types of devices on Key parameters including transconductance (Gm), field effect mobility (μFE), threshold voltage (Vth) and subthreshold slope (SS) were systematically analyzed. The doping distribution of the heavily doped epitaxial SiGe source/drain structure were subjected to more Coulomb scattering at cryogenic temperatures, whereas the doping distribution of the Schottky-barrier source/drain structure dictates that the device is mainly subjected to phonon scattering at cryogenic temperatures.
Open all abstracts, in this tab
Ghadah Abdulrahman AlZaidy and Hamdah T.A. Alanazi 2024 ECS J. Solid State Sci. Technol. 13 055006
Perovskite solar cells (PSC) have drawn interest in recent years due to their progressively improving power conversion efficiency (PCE), lightweight and wearable properties, straightforward solution fabrication process, suitability for flight, potential for deployment in ultra-lightweight space applications, and low-cost material constituents, among other factors. The efficiency of perovskite solar cells has exceeded 25% by developing novel low-cost synthesis methods and advancements in interface and electrode materials, enhancing the production of high-quality perovskite films. Moreover, perovskite solar cells' stability has been the focus of several studies. This review primarily examines recent advances in perovskite solar cells concerning their properties, composition, and synthesis methods. The main focus is to study transition metal oxide (TMO)-based nanocomposites for various PSC layers, including electron transport layers (ETLs), hole transport layers (HTLs), and other layers. These TMO-based nanocomposites were employed in perovskite solar cells, considering their band gap, carrier mobility, transmittance, and other relevant factors. The prospects of different TMO (iron, titanium, copper, nickel, etc) -based perovskite solar cells and their potential for commercialization feasibility have also been examined.

Highlights
The history of perovskites in solar cells and the properties of PSCs are discussed.
Strategies for large scale production of PSCs are the focus of this review.
Advances in TMO-based nanocomposites for various PSCs materials (Hole transport layer, electron transport layer, etc) are discussed in detail.
Lastly, the current problem in use of TMO-based nanocomposites for various PSCs materials is highlighted, and a future perspective is given to provide researchers with a pathway for further research.
Anita Gupta et al 2024 ECS J. Solid State Sci. Technol. 13 047006
The main characteristics of a good pH detecting system are higher sensitivity, ease of manufacturing process, and a micro-system. Ion sensitive field effect transistors (ISFETs), which are frequently employed as biosensors, offer significant advantages, and have gained prominence in various sectors. This review has highlighted the factors influencing sensitivity in pH sensing and explored various methods to enhance the sensor's sensitivity and overall performance. Miniature sensors play a crucial role, especially in industries, biomedical and environmental applications. For accurate pH measurements in both in-vivo and in-vitro systems, as well as for the device's miniaturization, the reference electrode (RE) must be positioned precisely in an ISFET device, considering both the device's physical dimensions and the distance between the sensing surface and the RE. Hence, this review provides valuable insights into the importance of sensitivity, miniaturization, and the role of the RE in ISFET devices, contributing to the advancement and application of pH sensing technology in diverse fields.
Himanshu Prasad Mamgain et al 2024 ECS J. Solid State Sci. Technol. 13 043010
Corrosion is an undesirable electrochemical reaction that leads to material degradation and affects material properties like ductility, malleability, conductivity, etc. The consequences of corrosion are machine failure, bridge failures, buildings collapse, and significant economic losses to GDP (4-5%). Furthermore, corrosion can pose serious safety risks that result in casualties which makes minimizing the effect of corrosion a great challenge. Traditional solutions like inhibitors, design modification, and paints are available to prevent corrosion but have many limitations, such as cost, durability, stability issues, and general inefficiency. In this context, a nanostructured superhydrophobic coating (SH) is gaining attention for its corrosion prevention efficiency and other broad industrial applications. The nano air pockets present in SH coating exhibit a high contact angle due to their unique combination of high surface roughness, distinctive nanostructure, and reduced surface energy. This reduces the surface area of between the corrosive substance,water droplet and the metal surface, leading to improved efficiency in resisting corrosion. In this paper, the recent advancement in electrodeposition to develop corrosion-resistant SH coatings on copper substrate and compression with other metals with their physical, chemical, and thermal stabilities are discussed. In many papers, scientists observed different types of surface morphology, texture, and surface energy, which give different tendencies to prevent surfaces from corrosion are also disscused . The constraints in fabrication and the prospects of the coating are also highlighted.
Highlights
An overview of the applications of copper and the problem of corrosion, factors affecting corrosion, and its impact in different industries.
A broad overview of rudiments of the superhydrophobicity
Detailed analysis of fabrication of SHCs for metal protection from corrosion by electrodeposition on copper and comparisons with other metals.
Other industrial applications of corrosion-resistant superhydrophobic coating are included.
Stability, conclusion, and future perspectives in fabricating superhydrophobic coating to minimize corrosion.
Bakr Ahmed Taha et al 2024 ECS J. Solid State Sci. Technol. 13 047004
Early diagnosis through noninvasive tools is a cornerstone in the realm of personalized and medical healthcare, averting direct/indirect infection transmission and directly influencing treatment outcomes and patient survival rates. In this context, optical biochip breathomic sensors integrated with nanomaterials, microfluidics, and artificial intelligence exhibit the potential to design next-generation intelligent diagnostics. This cutting-edge tool offers a variety of advantages, including being economical, compact, smart, point of care, highly sensitive, and noninvasive. This makes it an ideal avenue for screening, diagnosing, and prognosing various high-risk diseases/disorders by detecting the associated breath biomarkers. The underlying detection mechanism relies on the interaction of breath biomarkers with sensors, which causes modulations in fundamental optical attributes, such as surface plasmon resonance, fluorescence, reflectance, absorption, emission, phosphorescence, and refractive index. Despite these remarkable attributes, the commercial development of optical biochip breathomic sensors faces challenges, such as insufficient support from clinical trials, concerns about cross-sensitivity, challenges related to production scalability, validation issues, regulatory compliance, and contrasts with conventional diagnostics. This perspective article sheds light on the cutting-edge state of optical breathomic biochip sensors for disease diagnosis, addresses associated challenges, proposes alternative solutions, and explores future avenues to revolutionize personalized and medical healthcare diagnostics.
Madhu Bala and Sushil Bansal 2024 ECS J. Solid State Sci. Technol. 13 047003
Plant leaf disease identification is a crucial aspect of modern agriculture to enable early disease detection and prevention. Deep learning approaches have demonstrated amazing results in automating this procedure. This paper presents a comparative analysis of various deep learning methods for plant leaf disease identification, with a focus on convolutional neural networks. The performance of these techniques in terms of accuracy, precision, recall, and F1-score, using diverse datasets containing images of diseased leaves from various plant species was examined. This study highlights the strengths and weaknesses of different deep learning approaches, shedding light on their suitability for different plant disease identification scenarios. Additionally, the impact of transfer learning, data augmentation, and sensor data integration in enhancing disease detection accuracy is discussed. The objective of this analysis is to provide valuable insights for researchers and practitioners seeking to harness the potential of deep learning in the agricultural sector, ultimately contributing to more effective and sustainable crop management practices.
Open all abstracts, in this tab
Y. Kim et al 2024 ECS J. Solid State Sci. Technol. 13 054007
Ru deposition in advanced technology nodes can improve performance by providing low resistance in nanoscale features. In this study, we reported the electrochemical reactions of Ru3+ on an Ru surface using multi-cyclic voltammetry (CV) and behavior of additives during Ru electrodeposition using linear sweep voltammetry (LSV) and potentiostatic measurements. Disodium 3,3'-dithiobis(1-propanesulfonate) (SPS), polyvinylpyrrolidone (PVP), and a bromide ion (Br−) were added for bottom-up filling. We investigated the suppression behavior of PVP. The current density and the onset potential of suppressor breakdown were affected by the suppressor concentration. PVP interacted synergistically with Br− and showed additional suppression. PVP formed a suppressing layer with Br− after formation of a bromide layer. SPS could reduce roughness during electrodeposition of Ru. Based on these results, nano-trenches were filled with Ru using the optimized additive condition.
Bakr Ahmed Taha et al 2024 ECS J. Solid State Sci. Technol. 13 047004
Early diagnosis through noninvasive tools is a cornerstone in the realm of personalized and medical healthcare, averting direct/indirect infection transmission and directly influencing treatment outcomes and patient survival rates. In this context, optical biochip breathomic sensors integrated with nanomaterials, microfluidics, and artificial intelligence exhibit the potential to design next-generation intelligent diagnostics. This cutting-edge tool offers a variety of advantages, including being economical, compact, smart, point of care, highly sensitive, and noninvasive. This makes it an ideal avenue for screening, diagnosing, and prognosing various high-risk diseases/disorders by detecting the associated breath biomarkers. The underlying detection mechanism relies on the interaction of breath biomarkers with sensors, which causes modulations in fundamental optical attributes, such as surface plasmon resonance, fluorescence, reflectance, absorption, emission, phosphorescence, and refractive index. Despite these remarkable attributes, the commercial development of optical biochip breathomic sensors faces challenges, such as insufficient support from clinical trials, concerns about cross-sensitivity, challenges related to production scalability, validation issues, regulatory compliance, and contrasts with conventional diagnostics. This perspective article sheds light on the cutting-edge state of optical breathomic biochip sensors for disease diagnosis, addresses associated challenges, proposes alternative solutions, and explores future avenues to revolutionize personalized and medical healthcare diagnostics.
Sangeeta Palekar et al 2024 ECS J. Solid State Sci. Technol. 13 027004
The pursuit of rapid diagnosis has resulted in considerable advances in blood parameter sensing technologies. As advances in technology, there may be challenges in equitable access for all individuals due to economic constraints, advanced expertise, limited accessibility in particular places, or insufficient infrastructure. Hence, simple, cost efficient, benchtop biochemical blood-sensing platform was developed for detecting crucial blood parameters for multiple disease diagnosis. Colorimetric and image processing techniques is used to evaluate color intensity. CMOS image sensor is utilized to capture images to calculate optical density for sensing. The platform is assessed with blood serum samples, including Albumin, Gamma Glutamyl Transferase, Alpha Amylase, Alkaline Phosphatase, Bilirubin, and Total Protein within clinically relevant limits. The platform had excellent Limits of Detection (LOD) for these parameters, which are critical for diagnosing liver and kidney-related diseases (0.27 g dl−1, 0.86 IU l−1, 1.24 IU l−1, 0.97 IU l−1, 0.24 mg dl−1, 0.35 g dl−1, respectively). Machine learning (ML) algorithms were used to estimate targeted blood parameter concentrations from optical density readings, with 98.48% accuracy and reduced incubation time by nearly 80%. The proposed platform is compared to commercial analyzers, which demonstrate excellent accuracy and reproducibility with remarkable precision (0.03 to 0.71%CV). The platform's robust stability of 99.84% was shown via stability analysis, indicating its practical applicability.
V. I. Nikolaev et al 2023 ECS J. Solid State Sci. Technol. 12 115001
The properties of orthorhombic κ-Ga2O3 films grown by Epitaxial Lateral Overgrowth (ELOG) were studied by Scanning Transmission Electron Microscopy (STEM), X-ray diffraction, capacitance-voltage profiling, Microcathodoluminescence (MCL) spectroscopy and imaging. ELOG mask was formed by deposition of SiO2 stripes on TiO2 buffer prepared on basal plane sapphire, with the stripes going along the [11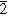 0] direction of sapphire. κ-Ga2O3 ELOG growth was performed using Halide Vapor Phase Epitaxy (HVPE), with ELOG wing of the structure formed by lateral overgrowth over the 20 μm-wide SiO2 stripes, while growth in between the stripes proceeded initially by vertical growth in the 5-μm-wide windows. TEM analysis showed that the material in the windows comprised 120o rotational nanodomains typical of κ-Ga2O3, while, in the wing regions, the material was single-domain monocrystalline. The films were conducting, with the net donor density close to 1013 cm−3. The data suggested the material in the windows have much higher resistance than in the wings. MCL spectra and imaging revealed much higher density of nonradiative recombination centers in the windows than in the wings.
0] direction of sapphire. κ-Ga2O3 ELOG growth was performed using Halide Vapor Phase Epitaxy (HVPE), with ELOG wing of the structure formed by lateral overgrowth over the 20 μm-wide SiO2 stripes, while growth in between the stripes proceeded initially by vertical growth in the 5-μm-wide windows. TEM analysis showed that the material in the windows comprised 120o rotational nanodomains typical of κ-Ga2O3, while, in the wing regions, the material was single-domain monocrystalline. The films were conducting, with the net donor density close to 1013 cm−3. The data suggested the material in the windows have much higher resistance than in the wings. MCL spectra and imaging revealed much higher density of nonradiative recombination centers in the windows than in the wings.
Younghyun You et al 2023 ECS J. Solid State Sci. Technol. 12 075009
WS2 is an emerging semiconductor with potential applications in next-generation device architecture owing to its excellent electrical and physical properties. However, the presence of inevitable surface contaminants and oxide layers limits the performance of WS2-based field-effect transistors (FETs); therefore, novel methods are required to restore the pristine WS2 surface. In this study, the thickness of a WS2 layer was adjusted and its surface was restored to a pristine state by fabricating a recessed-channel structure through a combination of self-limiting remote plasma oxidation and KOH solution etching processes. The reaction between the KOH solution and WOX enabled layer-by-layer thickness control as the topmost oxide layer was selectively removed during the wet-etching process. The thickness of the WS2 layer decreased linearly with the number of recess cycles, and the vertical etch rate was estimated to be approximately 0.65 nm cycle−1. Micro-Raman spectroscopy and high-resolution transmission electron microscopy revealed that the layer-by-layer etching process had a nominal effect on the crystallinity of the underlying WS2 channel. Finally, the pristine state was recovered by removing ambient molecules and oxide layers from the surface of the WS2 channel, which resulted in a high-performance FET with a current on/off ratio greater than 106. This method, which provides a facile approach to restoring the pristine surfaces of transition-metal dichalcogenide (TMDC) semiconductors with precise thickness control, has potential applications in various fields such as TMDC-based (opto)electronic and sensor devices.
Open all abstracts, in this tab
Dahiya et al
In recent years, there has been notable exploration and investigation of graphene nanocomposites (GNCs) through experimental, numerical, and computational methods. GNCs have gained attention due to their remarkable mechanical and thermal properties, particularly when Gr has been utilized as the reinforcing material. Gr, a two-dimensional material, possesses exceptional properties, including greater elastic modulus, thermal conductivity, and electrical conductivity. As a result, GNCs have emerged as promising materials for various applications in aerospace and automobiles. Computational techniques, including finite element method (FEM), molecular dynamics, and Monte Carlo analysis have been utilized to analyse different aspects of GNC. Among these, FEM stands out for designing and evaluating the mechanical properties of GNC, enabling researchers to simulate and analyse the characteristics of GNC structures under diverse loading conditions, optimizing their design and predicting mechanical performance. This review emphasizes the significance of Gr in various matrices, discusses the present cutting-edge status of FEM methodologies for Gr reinforcement, and highlights its advantages and purposes. Furthermore, it explores the governing parameters affecting the mechanical properties of GNC and briefly presents the different mechanical properties of NC. We also outline future research directions and potential applications of GNC for advancing future generations of materials.
D et al
The unique properties of light underlie the perspectives of quantum photonic technologies, optical interconnects, and a wide range of new sensors.Some of the most dangerous and deadly diseases are tumors, cancers, and brain lesions, which are expensive to detect and treat. Therefore, a low-cost and accurate method to diagnose them can prevent and treat the progress of this disease. In this work, we used the precise topological valley photonic crystal (TVPC) method for detection. TVPC is an important method for transmitting and controlling light in the optical device. The valley-spin locking in the topology state provides robust transfer and low propagation loss at the desired path.In order to design, by finding the edge band within the first bulk band gap of 146.4THz-155.9THz, the model and design of the topological ring resonator (TRS)were realized. We proposed a topological biosensorbyusing the hexagonal lattice air holes in the silicon slab with a compact size of 22.95µm×10µm. the quality factor and sensitivity at best value are2.905×104and9021nm/RIU respectively. This design can be implemented on the Complementary Metal-Oxide-Semiconductor (CMOS) technology as a high-sensitivity optical device.
Aslam et al
Amorphous indium gallium zinc oxide (a-IGZO) has recently made significant advancement as a key material for electronic component design owing to its compatibility with complementary metal oxide semiconductor technologies. A comprehensive analysis of reliability-related issues is required to determine the true potential of a-IGZO-based devices for next-generation electronics applications. To address this objective, we electrically characterize scaled-channel a-IGZO thin film transistors (TFTs) under positive bias (temperature) stress (PB(T)S). Both PBS and PBTS are characterized by positive and negative Vth shift, respectively, during the various gate stresses. In particular, the negative Vth shift is explained by the generation of donor-like traps stimulated by ionization of oxygen vacancy/hydrogen at elevated temperature. The TFTs exhibit relatively decent stability during the PBS operation. The analysis of devices with variant channel dimensions implies that long-channel devices exhibit relatively higher stability and performance compared to the short-channel ones. We also observe that the Vth can be controllably adjusted by employing the top gate (TG) with bottom gate sweep. Moreover, the stress-induced partial recovery mechanism is experimentally observed owing to detrapping of charges. Generally, the reported results infer a perceptive understanding of scaled-channel a-IGZO-TFTs which helps with shaping performance-enhancement strategies.
Rehman et al
Here an approach is presented to electrically operate the quantum tunnelling probability by exploiting the transported carriers at the interface of p-AlGaN/n-AlGaN/n++-AlGaN tunnel junction (TJ) with moderate Si and Mg-doping levels and optimized thickness with the help of simulation study. The simulation results show that the Augur recombination rate is successfully suppressed and quite a high radiative recombination rate is achieved in the 284 nm N-polar AlGaN-based TJ UV-B LEDs, which is attributed to the improved hole injection toward the MQWs when compared to C-LED (conventional-LED). It is found that C-LED has a maximum IQE (internal quantum efficiency) of 40% under 200 A/cm2 injection current with an efficiency drop of 15%, while the TJ-LED has a maximum IQE of 93% with an efficiency droop of 0%. In addition, TJ-based AlGaN LED emitted power has been improved by 6 times compared to the C-LED structure. This is attributed to the lower Augur recombination rate in the MQWs of N-AlGaN-based TJ UV-B LED. The operating voltages were reduced from 5.2 V to 4.1 V under 200 mA operation, which is attributed to the thickness and doping optimization in TJ and better selection of relatively lower Al-content in the contact layer
Zhu et al
Passivated emitter and rear contact (PERC) solar cells possess the highest photovoltaic market share at present. In industrial production, blistering of the rear silicon nitride (SiNx) passivation layer significantly affects the yield. In order to solve this problem, the relevant processes for manufacturing the PERC solar cells have been carefully studied. It was found that polishing of the silicon wafer rear surface, aluminum (AlOx) thickness, and the deposition process of the SiNx layer will affect the blistering ratio. By optimizing the manufacturing process mentioned above, the blistering ratio of the PERC solar cells has been effectively suppressed. This work not only provides reliable technical support for the yield improvement of the PERC solar cells but also provides some useful reference for the tunnel oxide passivated contact (TOPcon) and back contact (BC) solar cell industrial manufacture
Trending on Altmetric
Open all abstracts, in this tab
Muhammad Aslam et al 2024 ECS J. Solid State Sci. Technol.
Amorphous indium gallium zinc oxide (a-IGZO) has recently made significant advancement as a key material for electronic component design owing to its compatibility with complementary metal oxide semiconductor technologies. A comprehensive analysis of reliability-related issues is required to determine the true potential of a-IGZO-based devices for next-generation electronics applications. To address this objective, we electrically characterize scaled-channel a-IGZO thin film transistors (TFTs) under positive bias (temperature) stress (PB(T)S). Both PBS and PBTS are characterized by positive and negative Vth shift, respectively, during the various gate stresses. In particular, the negative Vth shift is explained by the generation of donor-like traps stimulated by ionization of oxygen vacancy/hydrogen at elevated temperature. The TFTs exhibit relatively decent stability during the PBS operation. The analysis of devices with variant channel dimensions implies that long-channel devices exhibit relatively higher stability and performance compared to the short-channel ones. We also observe that the Vth can be controllably adjusted by employing the top gate (TG) with bottom gate sweep. Moreover, the stress-induced partial recovery mechanism is experimentally observed owing to detrapping of charges. Generally, the reported results infer a perceptive understanding of scaled-channel a-IGZO-TFTs which helps with shaping performance-enhancement strategies.
Sergei Pavlovich Stepanoff et al 2024 ECS J. Solid State Sci. Technol.
Understanding the single event effects (SEE) sensitivity of microelectronic devices and circuits is essential for long-term mission success in ionizing radiation environments. SEEs occur when a single ionizing particle strikes a device with enough energy to cause anomalous malfunction or even a catastrophic failure event. It is conventionally viewed as an electrical phenomenon, whereas this study investigates the possible role of multi-physics. Specifically, we show that localized mechanical stress in electronic devices significantly impacts the degree of SEE sensitivity. We present a technique that indirectly maps both electrical and mechanical field localization to spatially map SEE sensitivity without any need for radiation test sources. It is demonstrated on the operational amplifier LM124 under both pristine and stressed conditions. To validate our hypothesis, our experimental results are compared with those obtained from the well-established pulsed-laser SEE technique. Excellent agreement between these results support our hypothesis that SEE susceptibility may have fundamental roots in both electrical and mechanical fields. Therefore, the ability to map the localizations in these fields may indirectly map the SEE sensitivity of large area electronics, which is very expensive in time and resources.
Yi-Wen Lin et al 2024 ECS J. Solid State Sci. Technol. 13 055004
This study reports a self-induced ferroelectric Ge-doped HfO2 (Ge:HfO2) thin film through interface reactions. In the first experiment, three treatments for forming interfacial layer (IL) were discussed through TiN/2-nm-thick Al2O3/2-nm-thick Ge:HfO2/GeO2/Ge metal-ferroelectric-insulator-semiconductor capacitors. The remnant polarization (Pr), leakage current, and interface trap density (Dit) were compared to select the most appropriate IL treatment. The results show that the in-situ ozone treatment under the standard atomic layer deposition process had the second highest 2Pr value as well as lower Dit values. Next, the thicknesses of Al2O3/Ge:HfO2 would be changed to 4 nm/2 nm and 3 nm/3 nm to investigate the ferroelectricity and leakage current. Although the 3-nm-thick Al2O3/3-nm-thick Ge:HfO2 shows a lower 2Pr value, the leakage current is much lower than 2-nm-thick Al2O3/2-nm-thick Ge:HfO2. The self-induced ferroelectric 3-nm-thick Ge:HfO2 thin film was then applied to fabricate Ge stacked nanowires gate-all-around field-effect transistor. The results show a steep subthreshold slope of 58 mV/dec for pFET and on-off current ratio > 105 and have high potential in low-power IC applications.
Wei-Chia Chen et al 2024 ECS J. Solid State Sci. Technol. 13 055003
This study investigated threshold voltage (VTH) instability in a Schottky p-GaN gate AlGaN/GaN high-electron-mobility transistor (HEMT) by using the double pulse test (DPT) with a 1 μs pulse width in the ON-state and OFF-state. OFF-state drain biases (VDS,OFF) of 100–400 V and ON-state drain currents of ID,ON 1–16 A were applied in the DPT to observe the post-DPT VTH shift. The ON-state currents did not strongly influence the device’s characteristics after the DPT. However, the OFF-state voltages, particularly VDS,OFF = 100 and 200 V, exerted notable effects. A TCAD simulation was conducted to investigate the mechanism underlying the VTH shift after the DPT at various VDS,OFF and ID,ON levels.
Iris Denmark et al 2024 ECS J. Solid State Sci. Technol.
Two approaches have been utilized to optimize the energy storage characteristics of doped carbon materials derived from Lignosol, a biomass product, to address the rising energy demand issues. Herein, phosphorus and nitrogen co-doped carbon (PNDC) materials with varying doping agent volumes were synthesized by utilizing microwave irradiation. Chemical activation and physical activation were employed to enhance these materials’ characteristics. Chemical activation was performed in a one-pot, single-step process, rather than a traditional multi-step protocol, using small amounts of potassium hydroxide. Furthermore, the physical activation method required multiple steps: doped carbon was prepared via microwave, exposed to water, filtered, frozen and then dried. With this, the expansion properties of water at freezing temperatures were exploited to alter the materials’ surface characteristics. All materials were characterized and compared for their physicochemical properties. All defect ratios supported the presence of doping. Additional results revealed that both chemical and physical activation approaches effectively modify the topographical features as well as the electrochemical activity (charge storage) of the doped carbon materials. The chemically activated doped carbon exhibited the highest resulting surface area of 1352 m2/g and a specific capacitance value of 347 F g-1 with excellent cycling stability as compared to other similarly synthesized materials.
Hui-Hsuan Li et al 2024 ECS J. Solid State Sci. Technol. 13 053008
We have developed a method that uses a half-cycle Hf precursor adsorption to subtly dope GeO2 IL of the Hf-based gate stack through in situ plasma-enhanced atomic layer deposition. This technique can effectively reduce GeO vaporization and improve the thermal stability of the GeO2 layer. Our results indicated that the accumulation capacitance (Cacc) undergoing higher temperatures showed no noticeable increase in the capacitance-voltage (CV) curves once Hf was delicately introduced into the GeO2 layer. According to the Ge 3d spectra of X-ray photoelectron spectroscopy, we found that the IL had a signal from extra Hf-O bonds; thus, we conclude GeO evaporation can be suppressed substantially by Hf incorporation. As a result, adding metal into GeOx IL to form HfGeOx achieved a remarkably low leakage current of 9 × 10−5 A cm−2 and the lowest interface trap density (Dit) of approximately 2 × 1011 eV−1 cm−2 at 500 °C of PMA. In addition, applying this gate stack structure to device fabrication significantly reduced the leakage current of the off-state and improved the effective peak hole mobility.
Jose Lopez Ninantay et al 2024 ECS J. Solid State Sci. Technol. 13 054004
The catalytic decomposition of poly(phthalaldehyde) with a photoacid generator can be used as dry-develop photoresist, where the exposed film depolymerizes into small molecules to allow the development of features via controlled vaporization. Higher temperatures enabled shorter dry-development times, but also promoted faster photoacid diffusion that compromised pattern fidelity. Trihexylamine was used as a base quencher to counteract acid diffusion in a phthalaldehyde-propanal co-polymer photoresist. The propanal co-monomer in the polymer improves the vaporization rate because it has a higher vapor pressure than phthalaldehyde. Addition of the base quencher was found to improve the contrast, pattern fidelity, and ease-of-handling of the dry-develop resist in a direct-write UV lithography tool. The dry-development of 4 μm features was achieved with no appreciable residue. For large area features, a spatially variable exposure method was used to direct the residue away from the exposed area. The gradient exposure method was used to produce 100 μm features. Plasma etching after dry-development was also used to achieve residue-free dry-developed patterns. These results show the benefits of incorporating base additives into a dry-develop depolymerizable resist system and highlight the need for addressing residue formation.
Hui-Hsuan Li et al 2024 ECS J. Solid State Sci. Technol. 13 055001
We propose a continuous fabrication method for HfO2-based gate stacks on a Ge bulk p-type metal–oxide–semiconductor capacitor (pMOSCAP) with HfGeOx interfacial layer by H2 plasma treatment through in situ plasma-enhanced atomic layer deposition. The electrical characteristics showed that the proper hydrogen plasma treatment could obtain an aggressively scaled equivalent oxide thickness of approximately 0.55 nm and a relatively low gate leakage current of 8 × 10−4 A cm−2 under PMA 500 °C.
William Cheng-Yu Ma et al 2024 ECS J. Solid State Sci. Technol. 13 045003
This work explores the characteristics of ferroelectric thin-film transistors (FeTFTs) utilizing an asymmetric dual-gate (DG) structure in both single-gate (SG) and DG operation modes. In the transfer characteristics, DG mode exhibits a memory window (MW) of 1.075 V, smaller than SG mode’s MW of 1.402 V, attributed to the back-gate bias effect causing a reduction in the device’s threshold voltage. However, DG mode demonstrates superior endurance characteristics with 106 cycles compared to SG mode’s 105 cycles. Additionally, the increase in erase pulse voltage (VERS) exacerbates the polycrystalline-silicon channel lattice damage of FeTFT, resulting in subthreshold swing (SS) degradation. Nevertheless, the extent of SS degradation from DG mode operation is significantly lower than that of SG mode, contributing to the superior endurance of DG mode. The elevation of program pulse voltage (VPRG) induces imprint and charge-trapping effects in the top-gate ferroelectric dielectric, leading to reduced endurance. Due to the use of SiO2 as the back-gate dielectric in FeTFT, DG mode exhibits lower impacts of charge-trapping effects from the top-gate ferroelectric dielectric layer, resulting in better endurance compared to SG mode. The asymmetric DG structure provides greater tolerance in the selection of VPRG and VERS.
Yuga Osada and Takashi Yanagishita 2024 ECS J. Solid State Sci. Technol. 13 043007
Fe substrates with a depression pattern were anodized to obtain Fe oxide films with a nanohoneycomb structure and orderly arranged cylindrical pores of uniform size. Crystalline Fe oxide films could be obtained by the heat treatment of amorphous samples obtained by the anodization of Fe substrates, but the atmosphere during heat treatment had a significant effect on the surface structure and crystallinity of the resulting samples. The heat treatment of the anodized samples in air produced a crystalline Fe oxide film consisting of Fe2O3 and Fe3O4, but the nanohoneycomb structure could not be maintained above 400 °C because the Fe substrate was oxidized during the heat treatment, and its surface structure changed significantly. On the other hand, the heat treatment of the anodized samples in N2 atmosphere yielded Fe3O4 nanohoneycombs, which retained their regular honeycomb structure after heat treatment. The evaluation of the capacitor properties of the heat-treated samples showed that the properties differed markedly owing to the effects of the surface structure and crystallinity, with the sample heat-treated at 400 °C in N2 atmosphere with the largest specific capacitance. The Fe3O4 nanohoneycombs obtained in this study are expected to be useful as electrodes for high-capacity capacitors.